 The final volume of the diluted sample is 1000 µL (1 mL), and the concentration is 1/10 that of the original solution. Mixing 100 µL of a stock solution with 900 µL of water makes a 1:10 dilution. For a 1:100 dilution, one part of the solution is mixed with 99 parts new solvent. For example, a 1:10 dilution is a mixture of one part of a solution and nine parts fresh solvent. They are described as ratios of the initial and final concentrations. Serial dilutions are often performed in steps of 10 or 100. The initial concentration and target range needed determines the size and number of dilution steps required. Doing this several times results in a range of concentrations. The diluted sample is then used as the base solution to make an additional dilution. Serial Dilution of Bacterial culture / Stock solut.To perform a serial dilution, a small amount of a well-mixed solution is transferred into a new container, and additional water or other solvent * is added to dilute the original solution.What is the dilution factor, i.e, how many more times dilute is it than the original concentration?įinal Volume = Volume of Diluent (Distilled Water here) + 100 (Volume of Stock ) = 800 mLĭilution Factor = Final Volume / Volume of Stock Aliquotĭilution Factor is 8 or the stock is 1/8 times diluted. You do a dilution by combining 100 ml volume of NaCl plus 700 ml unit volumes of Distilled water. Serial Dilution Calculation: Problems and Solutions Tube 4 (Dilution 4) - Take 900uL of sterial water in tube 4 and add 100uL from Tube 3 - so tube 4 will have10^2 cells/mL or 100 cells/mL. Tube 3 (Dilution 3) - Take 900uL of sterial water in tube 3 and add 100uL from Tube 2 - so tube 3 will have10^3 cells/mL. Tube 2 (Dilution 2) - Take 900uL of sterial water in tube 2 and add 100uL from Tube 1 - so tube 2 will have10^4 cells/mL. Tube 1 (Dilution 1) - Take 900uL of sterial water in tube 1 and add 100uL of bacterial stock - so tube 1 will have10^5 cells/mL. 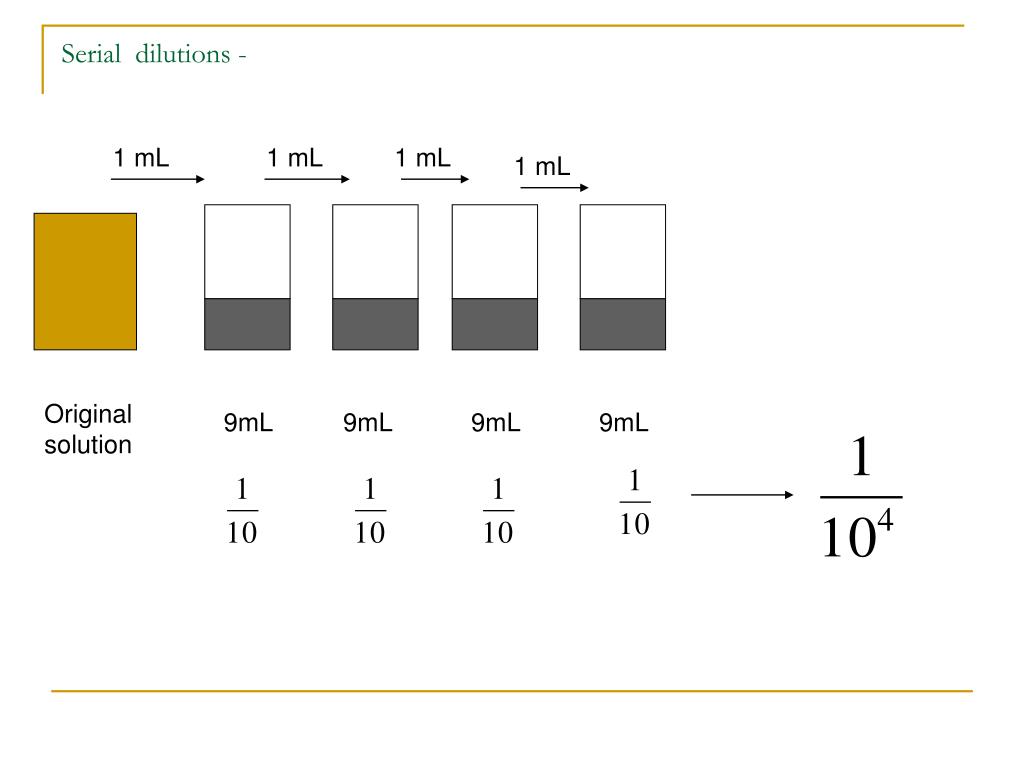
Stock of Bacterial Culture - 10^6 Bacterial Cells You need to do Four 10 fold dilution to a volume of 1mL to get 10^2 cells/mL final concentration. You have a bacterial culture, which has 10^6 bacterial cells and you want 100 cells/mL how to dilute the culture to get 100cells/mLįinal Concentration Required - 100 cells/mL or 10^2 cells/mL Serially dilute the bacterial culture, Bacterial culture is generally serially diluted for plating (for bacterial isolation), for re-inoculation or for other applications like checking optical density, etc. How to do Serial Dilution of Bacterial Culture?ĭilution of bacterial culture can be performed the same way as explained above. Stock solution is 1 M, after 1000 fold dilution it becomes - 1/1000 = 0.001 M. Label the tube as Dilution 1.įrom Dilution 1 tube take 10mL and mix 90mL of diluent and Label it as Dilution 2.įrom From Dilution 2 tube take 10mL and mix 90mL of diluent and Label it as Dilution 3.Īs we have done 10 fold dilutions, the tube 3 (Dilution 3) will be 1000 fold diluted than the stock. Mix 10mL of 1M stock with 90mL of Diluent. 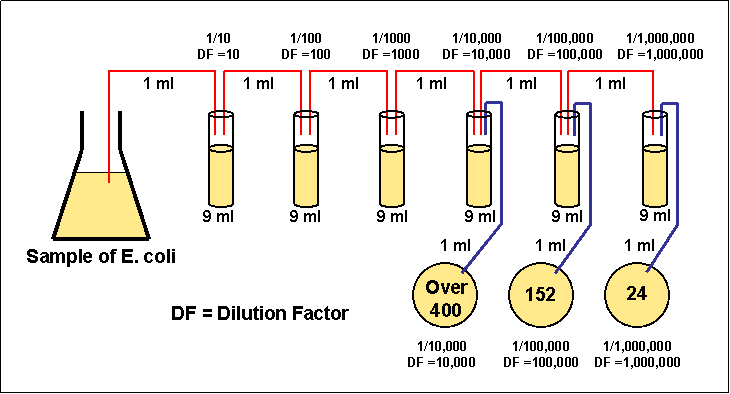
To do a 10 fold dilution of 1M Stock solution to 100mL, 
Since we need to do 10 fold dilution, dilution factor is 10.ĭiluent Volume required = 100mL - 10mL = 90mL. Lets see how to serially dilute 1 M stock solution to a final volume of 100 mL.ĭilution Factor = Final Volume / Volume of Stock. 10 fold dilution is actually 10^-1 dilution (Inverse of Dilution Factor: 1/10).ĭiluting a 1 M (Molar) Stock solution 1000 fold by serial Dilution.ġ000 fold serial dilution can be done by three 10 fold dilutions. Dilution is the inverse of Dilution Factor. The ratio of the Final volume to the aliqout volume of the stock is known as Dilution Factor (DF). To dilute a stock solution, Mix the stock solution with diluent. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
